Solid-state batteries promise more safety, higher energy density, and new degrees of freedom in cell design. However, the path from laboratory cells to industrial production is complex. Yet, laser processes can overcome central hurdles and enable breakthroughs.
Lithium-ion batteries are the standard for electrical energy storage – from consumer electronics to electromobility and stationary storage, and have undergone significant development in recent years.
However, the technology is hitting physical limits. Energy density is only growing slowly, safety remains limited due to liquid electrolytes, and dependence on critical raw materials such as nickel, manganese, or cobalt remains unresolved. Solid-state batteries are therefore considered the next generation of electrochemical storage. They promise higher energy densities through lithium-metal anodes, greater safety, and a wider temperature window thanks to solid electrolytes, as well as new degrees of freedom in cell construction.
However, they are still facing industrial maturity. Materials such as lithium-metal and sulfide-containing electrolytes require new process strategies, and production demands investments in specialized dry or inert gas environments. Here, laser technology can make crucial contributions, for example, through selective sintering of solid electrolytes, targeted structuring of interfaces, and contactless separation of ductile metals. Thus, it can prove to be a key technology on the path from laboratory cells to industrial solid-state batteries.
Potentials and Applications of Solid-State Batteries
Numerous manufacturers are currently advancing the development of solid-state cells. Asian companies like Toyota, BYD, Samsung SDI, and SVOLT have published ambitious timelines for pilot productions starting in 2027. European automakers like Mercedes-Benz and Stellantis are also testing initial semi-solid-state concepts together with partners, while Nissan is already building a pilot factory in Yokohama. These activities show: The technology is increasingly leaving the laboratory and approaching industrial implementation.
"The central advantage of solid-state batteries lies in their intrinsic safety," explains physicist Stoyan Stoyanov from the Separation group at the Fraunhofer Institute for Laser Technology ILT. "Since they do not rely on liquid electrolytes, the risk of leaks or thermally induced fire incidents is eliminated. Additionally, the high mechanical stability of many solid electrolytes inhibits the formation of lithium dendrites, which are the main cause of internal short circuits in conventional cells."
In addition to safety, the higher energy density primarily drives interest. Lithium-metal anodes with a specific capacity of 3860 mAh g⁻¹ far exceed graphite anodes. In combination with thin, solid electrolytes, this can achieve range and weight advantages, a crucial factor for electromobility and aviation.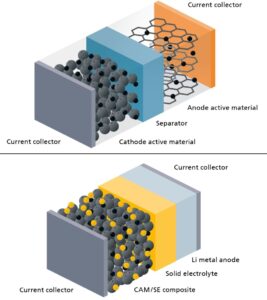
Initial application fields are emerging where maximum safety and performance are critical: in aerospace, motorsport, medical technology, or high-security data storage. Here, the higher energy density justifies the complex manufacturing.
For the mass market, economic competitiveness remains initially limited. The production infrastructure is under development, and established lithium-ion systems continue to evolve in parallel.
"Solid-state batteries will coexist alongside conventional lithium-ion cells for the foreseeable future and will primarily serve particularly demanding applications in the automotive industry, such as the luxury vehicle market," says Stoyanov.
Challenges in Manufacturing
As promising as the potentials of solid-state batteries are, the hurdles for industrial implementation are significant. Particularly, handling lithium-metal anodes poses high demands: The material is attractive due to its extraordinarily high specific capacity but proves to be extremely sensitive in processing. It reacts strongly with oxygen and moisture, easily forms passive layers, and can ignite under mechanical stress. Conventional cutting or rolling processes quickly reach their limits here.
Fundamental difficulties also arise on the side of solid electrolytes. Oxide ceramic materials such as lithium-lanthanum-zirconate (LLZO) must be sintered at around 1200 °C. This often leads to lithium losses and secondary phases that reduce ionic conductivity. Such losses are not only a technological but also an economic problem, as they render expensive raw materials unusable. While so-called sacrificial powders can partially compensate for these effects, the process remains complex and sensitive to the smallest fluctuations.
"Another bottleneck is the interface between electrolyte and anode. High interfacial resistances reduce performance and increase the risk of inhomogeneities during lithium plating and stripping. Mastering this interfacial chemistry is the basis for stable and durable cells," explains Florian Ribbeck from the High-Temperature Functionalization group at Fraunhofer ILT.
In addition to these material-specific aspects, the production conditions themselves pose a significant challenge. Solid-state batteries consistently require inert gas or dry room atmospheres, necessitating high investments in infrastructure. Initial analyses show that in industrial ramp-up, scrap rates of up to 30 percent can occur, leading to losses in the millions per day.
Even in established lithium-ion lines, the high scrap rate is an acute problem. In solid-state cells, this issue is exacerbated, as there are currently no closed recycling paths for the yet-to-be-standardized materials. Each defective prototype therefore means not only economic damage but also the loss of valuable raw materials. "Laser-based processes can help increase process stability and avoid scrap from the outset," says Ribbeck.
Laser Sintering of Solid Electrolytes
One research approach at Fraunhofer ILT is the processing of oxide ceramic solid electrolytes like LLZO. This material is considered particularly promising because it exhibits high electrochemical stability against lithium-metal anodes and is less reactive to environmental conditions compared to sulfide-containing electrolytes.
"At Fraunhofer ILT, we investigate how laser radiation can be used as a locally confined and highly dynamic energy source to selectively densify LLZO layers," explains Florian Ribbeck. "The advantage lies in the rapid heating while simultaneously controlling cooling. This can reduce lithium losses and avoid temperature incompatibilities within the cell assembly."
Initial experiments show homogeneous densifications, although crack formation and delaminations remain a central research topic. In addition to LLZO, NASICON-like electrolytes such as lithium-aluminum-titanium-phosphate (LATP) are being investigated, which have similar process requirements but different stability windows.
Laser structuring for better interfaces
In addition to the densification of the electrolyte layers, the quality of the interface to the lithium metal anode is crucial for the performance of solid-state cells. 'Here, high transition resistances often occur, which limit the electrochemical behavior,' explains Tim Rörig from the surface structuring group at Fraunhofer ILT about the challenge. 'Furthermore, the low wettability of the ceramic surfaces complicates a homogeneous lithium deposition.'
Rörig and Ribbeck are therefore investigating how the interfaces can be optimized through targeted laser structuring. Using ultrashort laser pulses in the femtosecond range, they introduce microstructures into the surface of the solid electrolyte. These structures increase the effective contact area and promote a more uniform distribution of current, potentially reducing the interfacial impedance. 'We have shown that reproducible structures can be generated in the range of around 30 µm,' explains Rörig.
However, the previous results also highlight the complexity of the interaction. While the structured surfaces showed improved wettability in some cases, the overall resistance of the cell sometimes increased. The researchers suspect that both changes in the crystal structure and process-related defects play a role.
Using Raman spectroscopy and other analytical methods, the researchers are currently characterizing the structural changes in the crystal lattice after laser processing. In parallel, they are investigating targeted Li plating to better control the contact, as well as concepts of so-called 'anodeless batteries,' where lithium is deposited only during the first charging process.
Laser cutting of lithium metal electrodes
Another focus at Fraunhofer ILT is on the separation of lithium metal foils for use as anode material. 'Lithium metal is considered a central component for the next generation of high-energy cells, but it poses significant challenges for manufacturing technology,' explains Stoyan Stoyanov. 'The material is soft, highly adhesive, and extremely reactive. Conventional mechanical methods such as rotary knives or punching quickly lead to smearing, tool sticking, and inhomogeneous cutting edges.' Furthermore, only linear cutting geometries can be realized mechanically, which severely limits flexibility in cell layout. Laser technology opens up new possibilities here. As a contactless and wear-free process, it allows for precise cuts and flexible contours.
However, both mechanical and laser-based processes require processing exclusively in closed inert gas or dry room atmospheres. These are essential for the safe handling of lithium but bring their own process engineering challenges. 'Argon is particularly suitable because it prevents oxidation and thus allows for uniform edges, but it is costly,' explains Stoyanov. 'Nitrogen is significantly cheaper, but it leads to the formation of lithium nitrides. Atmospheres with moisture promote oxides and hydroxides.' Such reaction products increase the energy demand of the process and can simultaneously degrade the electrochemical properties of the electrode.
Studies are already underway aimed at more cost-effective process environments that should better control the interactions at the lithium surface. 'However, these approaches are still in their infancy. In our own lab demonstrator, we therefore rely on a pure argon atmosphere with a dew point below -70 °C. Other gas environments can also be realized in principle.'
An additional challenge is to avoid particles and splashes that can occur during the laser process. They impair surface quality and lead to defects in the later cell assembly. Therefore, Stoyanov and his team are developing process strategies to deliberately control the ablation and efficiently remove emissions.
Ultrashort pulse lasers that operate with pulse durations in the picosecond range are an option to achieve high-quality cutting edges that are free from critical burr formation and have a minimal heat-affected zone. The team is also investigating technologically simpler to integrate and economically interesting options such as the use of nanosecond lasers, which allow for acceptable cutting quality at lower investment costs. In parallel, the researchers are working on concepts to integrate the laser processes into scalable production environments, for example, using compact mini-environments that can be specifically filled with inert gas.
Bridge to industrial implementation
The transfer of solid-state batteries from the lab to industrial production requires not only new materials but also, or especially, robust processes. Here, the production of lithium-ion cells provides a valuable reference. Many process steps from electrode manufacturing to cell assembly to final processing are fundamentally comparable, although the requirements for solid-state cells are significantly higher.
Laser technologies are already established in lithium-ion manufacturing. They are used in laser slitting, i.e., the precise longitudinal cutting of electrode foils, in laser drying to quickly and energy-efficiently remove solvents, or in laser notching, the notching of current collectors. These experiences can largely be transferred to solid-state cells. However, the demands for precision, purity, and material stability increase significantly: even the smallest particles, defects, or chemical changes can impair the function of the cells.
'Therefore, laser processes are gaining further importance,' believes Stoyanov. 'Their contactless, selective energy input allows for highly precise processing that can be integrated into protected environments such as dry rooms or mini-environments. This makes the laser a tool that can meet both material requirements and take into account the strict environmental conditions.'
Thus, the process chains developed in the lab can be transferred into an industrial logic. Where high scrap rates and long lead times currently dominate, laser-based processes can make a decisive contribution to ensuring the scalability and economic viability of solid-state batteries.
Positioning of Fraunhofer ILT

The Fraunhofer Institute for Laser Technology ILT consolidates its expertise along the entire value chain of solid-state batteries. The focus is on laser-based manufacturing steps that are crucial for both material development and later industrialization. This includes laser sintering of solid electrolytes, laser structuring to optimize interfaces, laser cutting of lithium metal foils, and methods for contacting and integrating into the cell assembly.
While one group investigates the properties and limits of new electrolytes and anode materials, another team develops processes to process these materials reliably and scalably. 'This dual perspective allows us to bridge the gap between lab demonstration and industrial implementation early on,' summarizes Ribbeck.
Nevertheless, solid-state batteries will not quickly displace established lithium-ion cells, even though they open up new perspectives for applications that demand the highest standards of safety and energy density. 'Aerospace, medical technology, high-performance vehicles, or an uninterruptible power supply, the so-called UPS, for data centers and hospitals are examples where the advantages of solid electrolytes justify the additional effort,' says Stoyan Stoyanov. In the medium to long term, a move into broader markets could succeed with decreasing production costs.
For Europe, this presents a special opportunity. While the mass market for lithium-ion cells is strongly dominated by Asian manufacturers, there is no established industrial monopoly in the field of solid-state technology yet. Companies and research institutions can position themselves early, shape standards, and build new value chains.
Contact:





